Up to 13%, the upper limit of concentration for potassium dichromate, it has generally been thought that the behavior of the two dichromates is roughly the same at approximately the same concentrations, but recent research suggests that ammonium dichromate produces a better quality image (greater diffraction efficiency) even when the two dichromates are measured precisely to give the same number of chromium atoms per unit of colloid. I'll explain in greater detail why this is so on the chemistry page. And of course between 13% and 27%, ammonium dichromate is the only option, since potassium dichromate can't be mixed more concentrated than 13%.
The main issues to consider in deciding on dichromate concentration are speed and contrast. As dichromate concentration goes up, exposure time decreases linearly and contrast decreases (number of steps printed increases but the contrast is flatter). As dichromate concentration goes down, exposure time increases linearly and number of steps printed decreases, resulting in a contrastier print.
Here's a visual that shows the relationship between dichromate concentration, speed, and contrast, a strong relationship that has been established for decades; I didn't just discover it myself. This visual simply demonstrates a long- and well-established principle. I ran five tests at five different concentrations, but in the interest of space and of not boring you to death, I'll show only the two outside results. They show clearly that printing speed varies directly with dichromate concentration, and contrast varies indirectly with dichromate concentration. In other words, the more concentrated, the faster printing; the less concentrated, the more contrasty. I found a near-perfect linear relationship in the five tests between speed and concentration; half the concentration of dichromate took twice as long to print, everything else held constant; 1/3 the concentration took 3 times as long; 1/4 the concentration required 4 times the exposure; 1/5 the concentration, 5 times the exposure.
I used to think that saturated ammonium dichromate constituted the practical upper limit of advantage from increasing dichromate concentration, until I tried doubling the saturated dichromate in relation to the gum/pigment to print a heavily overpigmented mix (in other words, 2 units saturated ammonium dichromate: 1 unit gum/pigment) and found that the speed was again increased commensurately to the increase in dichromate. So I haven't yet found the upper limit where increase in dichromate fails to improve printing speed.

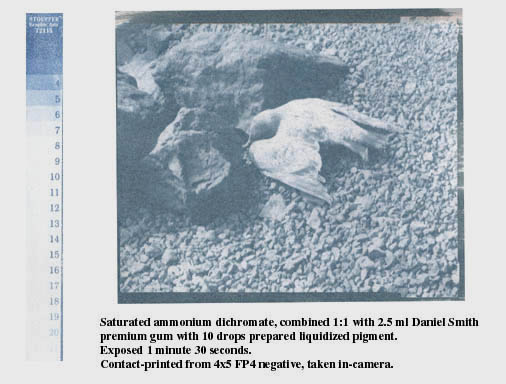
The test strip shows 8 separated steps; the 8th one may not be visible in the jpeg, but it's there in the actual test strip. Development time for this image was 30 minutes.
Below is the same pigment mix, same negative, but with the dichromate diluted to 1/5 of the saturated concentration:

This was printed at 7 minutes 30 seconds, exactly 5x the printing time for saturated dichromate. With any shorter exposure, the image simply washed away. There are four steps in the test strip that can be differentiated; steps 4 through 7. As you can see, besides being five times slower, the diluted dichromate prints with greater contrast (fewer steps).
I prefer saturated ammonium dichromate; I find the increased contrast of dilute dichromate unappealing and prefer the longer tonal scale and the greater speed of the saturated ammonium dichromate. But this is a matter of personal preference; by trial and error you'll find a dichromate concentration that suits you.
There is another perception (another myth, I'd call it) among some gum printers that potassium dichromate is "better" than ammonium dichromate, because ammonium dichromate has a tendency to pigment stain, or dichromate stain. I've printed with saturated ammonium dichromate exclusively, and have never had problems with either pigment stain or dichromate stain, so this assertion is inexplicable to me. I've talked to several people who hold this belief, and I've come to a tentative conclusion that the issue isn't staining per se, but speed. When a fast light is combined with a fast dichromate, it's easy to overexpose, which can result in dichromate stain or such a profoundly overexposed gum layer that it can resemble pigment staining, but isn't, as would become apparent if the print was left to develop for a longer time.
Light source: A secondary consideration in choosing a dichromate concentration is the light source you use. If you use a very intense light source, like a 3000 watt plateburner, or the sun, you may find that a more dilute concentration will work better for you; saturated ammonium dichromate may actually be too fast; in other words, even a very short exposure may result in overexposure. In the dim direct sun of the Pacific Northwest coast, however, I find that I can print a very nice print in the sun in 10-20 seconds depending on sun intensity.
Environmental considerations: Some gum teachers recommend a diluted dichromate out of consideration for the environment, but considering the miniscule amounts of dichromate we use, the environment is going to get along fine no matter which concentration of dichromate you choose, so I wouldn't make that an important part of your decisionmaking process. I use .675 grams of dichromate to make enough emulsion for a printing session of several prints; if I cut the dichromate to 1/5 as above, it's true that I would use less dichromate, but the difference between .675 grams and .135 grams is absolutely and totally insignificant as far the environment is concerned, and doesn't warrant giving up what I feel I would give up in speed and tonal subtlety to use a diluted dichromate. I've been told by two toxic waste experts that as long as I use such small amounts and as long as it goes through a sewage treatment plant, it poses no potential danger to the environment. The danger to the environment is when you're pouring barrelsful of it into the groundwater. (However, I wouldn't put it into a septic tank, even in these tiny amounts).
[Safety warning: Hexavalent chromium is a known carcinogen, when breathed. The greatest danger is to workers who spray automobile parts with chrome plating, and other applications that involve airborne chromium; these workers tend to develop cancers in the mouth and breathing passages at a significantly greater rate than the population as a whole. Our uses are much less dangerous, but I was warned by a chemist that I should always wear a mask when weighing and mixing the powder, and I will pass this warning on to you. Some people develop a contact dermatitis to the dichromate in solution, and the careful worker may want to wear gloves while handling the print in the dichromate-contaminated water. I don't do this because I just can't be bothered, but I may live to regret it some day. Also, dichromates can be fatal when swallowed, even in small amounts. There is an account in medical literature of a child who died after ingesting 1 gram of dichromate.]
Copyright Katharine Thayer, all rights reserved